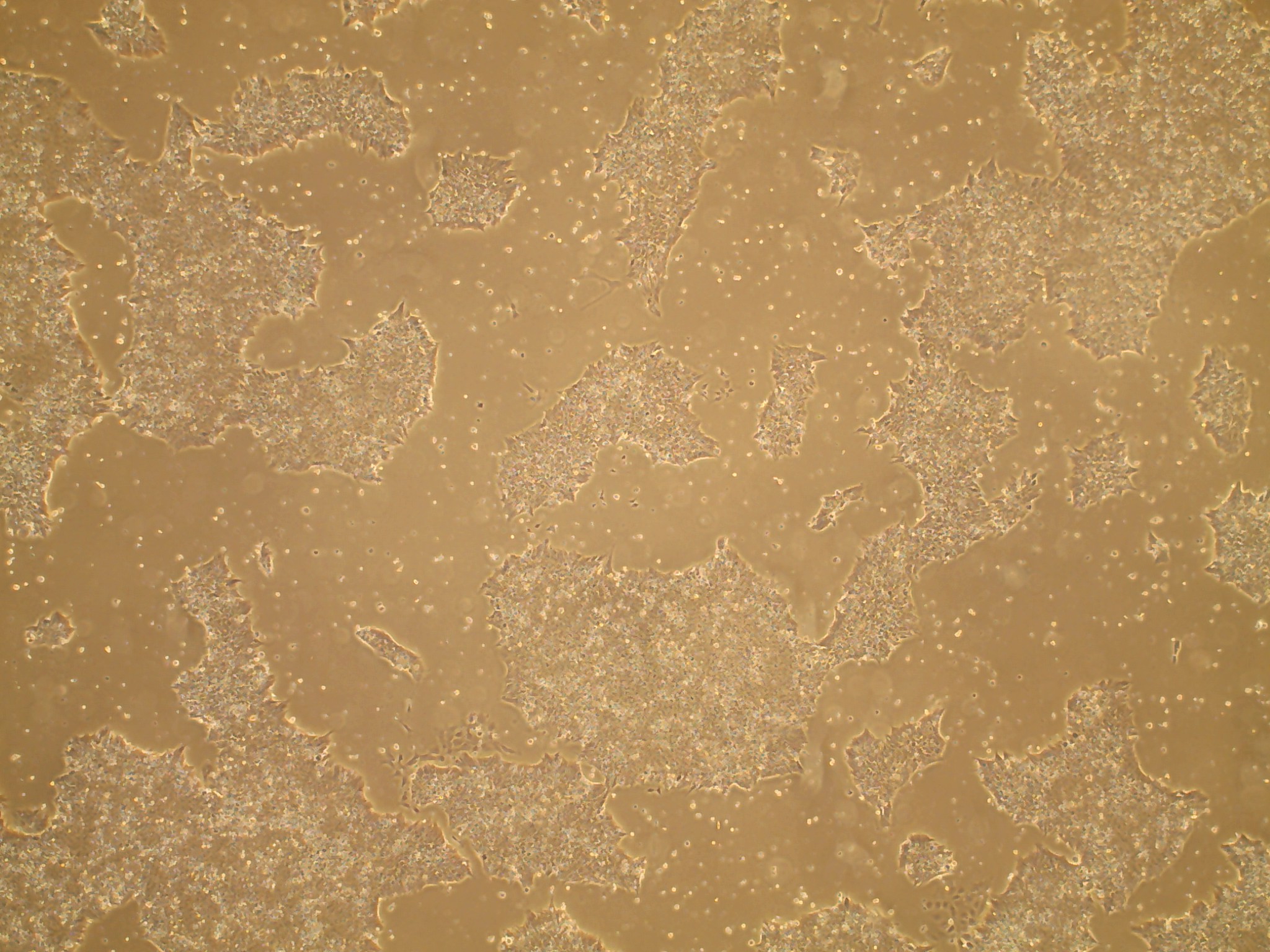
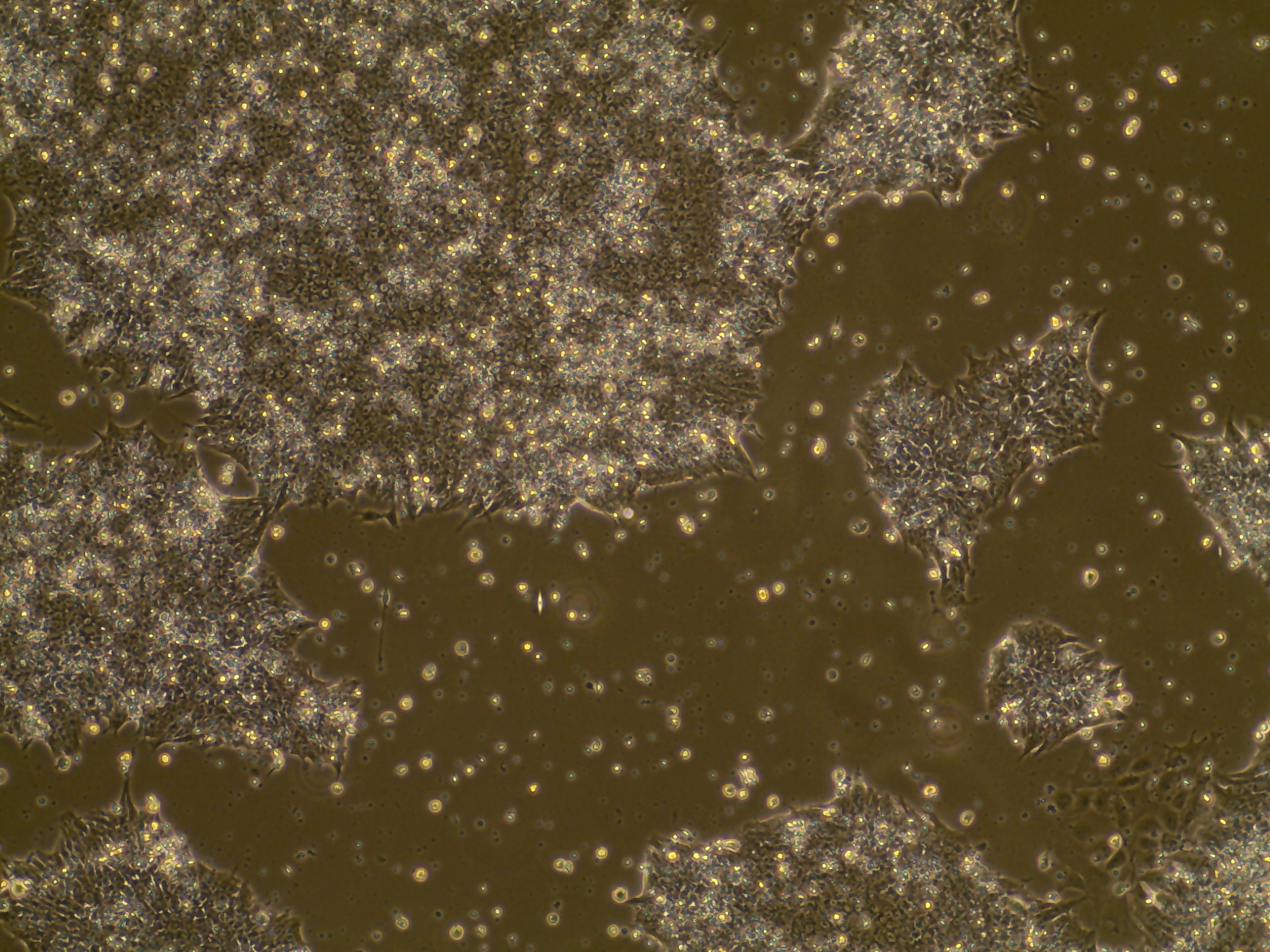
If the cells you would like to access are currently listed as unavailable or
you would like information on local partners in USA, Australia, New Zealand or Japan who can support order and delivery,
please get in touch via
Contact@EBiSC.org.
ESi002-A
SP08#1
iPSC line
A batch specific Certificate of Analysis will be available to
download once you receive your EBiSC iPSC line.
General#
Cell Line |
|
| hPSCreg name | ESi002-A |
| Alternative name(s) |
SP08#1
|
| Cell line type | Human induced pluripotent stem cell (hiPSC) |
| Similar lines |
EDi001-A-2 (AST23-1KO-3, AST22-1KO-3, AST-23_SCAKO Clone 3, AST-22_SNCAKO Clone 3) Donor's gene variants: SNCA, SNCA, SNCA, SNCA Donor diseases: Parkinson disease EDi001-A-3 (AST23_SNCAKO Clone 1, AST22-1KO-1, AST23-1KO-1, AST22_SNCAKO Clone 1) Donor's gene variants: SNCA, SNCA, SNCA, SNCA Donor diseases: Parkinson disease STBCi004-B-1 (SFC832-03-06 LRRK2WT/WT C47) Donor's gene variants: LRRK2 Donor diseases: Parkinson disease EDi001-A (AST22, AST23, SAMEA3319992) Donor's gene variants: SNCA, SNCA, SNCA Donor diseases: Parkinson disease |
Provider |
|
| Depositor | Spanish Stem Cell Bank (ES) |
| Distributors |
EBiSC
|
| Derivation country | Spain |
External Databases |
|
| hPSCreg | ESi002-A |
| BioSamples | SAMEA3303008 |
| Cellosaurus | CVCL_V202 |
| Wikidata | Q54955406 |
General Information |
|
| Publications | |
| This EBiSC line can be used for: |
Yes
Research use: allowed
Clinical use: no
Commercial use: no
|
Donor Information#
General Donor Information |
|
| Sex | female |
| Age of donor (at collection) | 65-69 |
Phenotype and Disease related information (Donor) |
|
| Diseases | A disease was diagnosed.
|
External Databases (Donor) |
|
| BioSamples | SAMEA3302974 |
hIPSC Derivation#
General |
|
| Source cell type |
An epithelial cell of stratified squamous tissues, including skin, oral mucosa (Hiroshima et al., 2011), and esophagus (Whelan et al., 2018), that produces keratin proteins and secretes antimicrobial peptides to form a resilient barrier against environmental damage, dehydration, pathogens, and microbial invasion. It undergoes successive stages of differentiation marked by changes in keratin expression, supporting tissue integrity, wound repair, and contributing to immune defense.
|
| Age of donor (at collection) | 65-69 |
Reprogramming method |
|
| Vector type | Integrating |
| Vector | Virus (Retrovirus) |
| Genes | |
| Is the used vector excisable? |
Unknown |
| Absence of reprogramming vector(s)? |
Unknown |
| Reprogramming vectors silenced? |
Yes |
| Methods used |
RT-PCR
|
Vector free reprogramming |
|
Other |
|
| Derived under xeno-free conditions |
No |
| Derived under GMP? |
No |
| Available as clinical grade? |
No |
Culture Conditions#
Latest released batch |
|
| Culture medium | mTeSR |
| Passage method | EDTA |
| Surface coating | Matrigel / Geltrex |
| O2 concentration | 21 |
| CO2 concentration | 5 |
| Temperature | 37 |
The following are the depositor culture conditions, they do not refer to any specific batch.
| Surface coating | Gelatin | |||||||||||||||
| Feeder cells |
Human foreskin fibroblasts Cellfinder Ont Id: CELDA_000001419 |
|||||||||||||||
| Passage method | Mechanically | |||||||||||||||
| O2 Concentration | 21 % | |||||||||||||||
| CO2 Concentration | 5 % | |||||||||||||||
| Medium |
Other medium:
Base medium: Knockout DMEM
Main protein source: Knock-out serum replacement Serum concentration: 20 % Supplements
|
Characterisation#
Analysis of Undifferentiated Cells
| Marker | Expressed | Immunostaining | RT-PCR | Flow Cytometry | Enzymatic Assay | Expression Profiles |
| Alkaline Phosphatase |
Yes |
|
||||
| NANOG |
Yes |
|
||||
| POU5F1 (OCT-4) |
Yes |
|
||||
| SSEA-3 |
Yes |
|
||||
| SSEA-4 |
Yes |
|
||||
| TRA 1-60 |
Yes |
|
||||
| TRA 1-81 |
Yes |
|
||||
| SOX2 |
Yes |
|
Differentiation Potency
In vivo teratoma
In vitro spontaneous differentiation
| Marker | Expressed |
| SMA |
Yes |
| CS |
Yes |
| SOX9 |
Yes |
In vivo teratoma
In vitro spontaneous differentiation
| Marker | Expressed |
| Tuj1 |
Yes |
| GFAP |
Yes |
Microbiology / Virus Screening |
|
| HIV 1 | Negative |
| HIV 2 | Negative |
| Hepatitis B | Negative |
| Hepatitis C | Negative |
| Mycoplasma | Negative |
Genotyping#
Karyotyping (Cell Line) |
|
| Has the cell line karyotype been analysed? |
Yes
|
Other Genotyping (Cell Line) |
|
| Is there genome-wide genotyping or functional data available? |
Yes
|